|
4/20/2023 0 Comments Boc deprotection 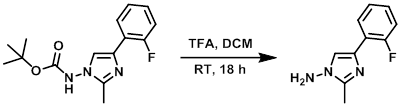
Even though this is not strictly an amine protection, the method can be useful in the design of the synthetic strategy. Dioxane and methanol have also been reported as solvents for the reaction, while an alternative common method is refluxing a biphasic mixture of chloroform and water, with sodium bicarbonate as a base.Ĭommon bases for the transformation are sodium hydroxide, 4-dimethylaminopyridine (DMAP) and sodium bicarbonate.Ī completely different route for the preparation of protected amines is the direct reaction of an alkyl halide with di-tert-butyl-iminodicarboxylate. It is typically performed in water, water/THF, THF, or acetonitrile at room temperature or moderate heat (40☌) in the presence of a base. The process usually achieves high yields and fast conversions under relatively mild conditions. The reaction conditions for the amine protection are quite flexible. The BOC (tert-butyloxycarbonyl) protecting group, chemically a di-tert-butyl dicarbonate (Boc2O), is probably the most common amine protecting group in non-peptide chemistry. Benzyl Protection and Deprotection of Alcoholsĭeprotection Amine Protection / Deprotection 1-PROTECTION.Methyl-Ester Protection and Deprotection.Mechanism Top Citations Related Reactions A mild procedure for the clay catalyzed selective removal of the tert-butoxycarbonyl protecting group from aromatic amines. Selective removal of the tert-butoxycarbonyl group from secondary amines: ZnBr2 as the deprotecting reagent. Selective synthesis of carbamate protected polyamines using alkyl phenyl carbonates. Pittelkow, M., Lewinsky, R., Christensen, J.B. Environmentally benign N- boc protection under solvent-and catalyst-free conditions. Jia, X., Huang, Q, Li, J., Li, S., Yang, Q. Montmorillonite K10 clay in ClCH2CH2Cl selectively cleaves aromatic N-BOC groups, leaving aliphatic N-BOC amines intact.ZnBr2 in CH2Cl2 selectively cleaves secondary N-BOC groups, leaving primary N-BOC groups untouched.One way to selectively protect primary amines in polyamines is to use t-BuOCO2Ph in CH2Cl2, or DMF.The BOC group can also be added without the use of a solvent.: Strong Organic Acid (TFA, p-Toluenesulfonic Acid), DCM Since both protection and deprotection reactions produce CO2 gas, closed systems should not be used. The protected amine is first protonated by TFA, triggering the production of a t-butyl cation and carbamic acid, which is decarboxylated to yield the amine. Trifluoroacetic acid (TFA) and dichloromethane (DCM) are commonly used to remove the BOC group. The base then abstracts a proton from the positively charged amine. The amine attacks a carbonyl site of BOC2O, creating a t-butyl carbonate leaving group that breaks down to carbon dioxide gas and t-butoxide. The BOC (tert-butoxycarbonyl) group is used to protect amines in synthetic reactions, is probably the most common amine protecting group in non-peptide chemistry.ĭi-tert-butyl dicarbonate, BOC2O (also known as di-tert-butyl pyrocarbonate or BOC anhydride), triethylamine (TEA), and tetrahydrofuran (THF) are commonly used to add the BOC group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
